Prueba CE/IVD qPCR Covid-19 en 2 horas

QuantVirus Real-Time PCR Coronavirus Test - ¡Detecta con precisión el SARS-CoV-2 en sujetos positivos en 2 horas!
Kit oficial para el diagnóstico de Covid-19 en Francia
La prueba de detección CE-IVD QuantVirus Real-Time PCR Coronavirus (SARS-CoV-2) es una prueba de diagnóstico in vitro basada en la tecnología de PCR en tiempo real, desarrollada para la detección específica del ARN viral del SARS-CoV-2 extraído de hisopos nasofaríngeos, hisopos orofaríngeos y esputo de pacientes sospechosos de estar infectados con el nuevo coronavirus. La sensibilidad analítica es de 1 copia por reacción (100 copias/mL) de ARN viral del SARS-CoV-2 con una confianza del 95%. El ensayo es altamente específico sin reacción cruzada con diferentes tipos de especies no SARS-CoV-2 analizadas.
Información clave del producto
- Autorización CE-IVD
- Fabricado en EE.UU.
- Detecta tres genes, Orf1ab, N y E
- Detecta diferentes cepas del virus SARS-CoV-2
- Detecta tan sólo 1 copia de ARN del virus
- Menos de 2 horas desde el ARN hasta los resultados en la mayoría de las máquinas qPCR
Tipo de muestra
Torundas nasofaríngeas, torundas orofaríngeas y esputo
Máquinas validadas
Thermo Fisher (ABI) QuantStudio 5, Thermo Fisher (ABI) 7500 Fast Dx y Bio-Rad CFX 384
Tamaño del paquete
Las pruebas de detección de coronavirus (SARS-CoV-2) son cruciales
El brote mundial del nuevo coronavirus, SARS-CoV2, causó la muerte de más de 3000 personas y enormes daños económicos. La cifra sigue aumentando. La detección precoz del virus es esencial para evitar que infecte a más personas. Sin embargo, en Estados Unidos, la falta de disponibilidad a tiempo de suficientes kits de detección fiables ha causado preocupación y pánico en la sociedad.
*Las cifras están calculadas a 9 de marzo de 2020 por https://www.worldometers.info/coronavirus/.
- Sin la prueba, el público en general no sabe si contrae el nuevo coronavirus o el virus de la gripe común, ya que comparten síntomas similares.
- Sin la prueba, el público en general no sabe si es portador del virus sin síntomas e infecta a sus allegados y a otras personas que estén en contacto con él.
- Sin la prueba, el público en general no sabe si es portador del virus y pierde la oportunidad de recibir tratamiento
Para satisfacer las necesidades médicas no cubiertas, DiaCarta ha desarrollado la Prueba de Detección de Coronavirus por PCR en Tiempo Real QuantVirus (SARS-CoV-2) para detectar con precisión el virus con alta sensibilidad utilizando hisopos nasofaríngeos, hisopos orofaríngeos y esputo de sujetos sospechosos.
¿Cómo funciona la prueba QuantiVirus?
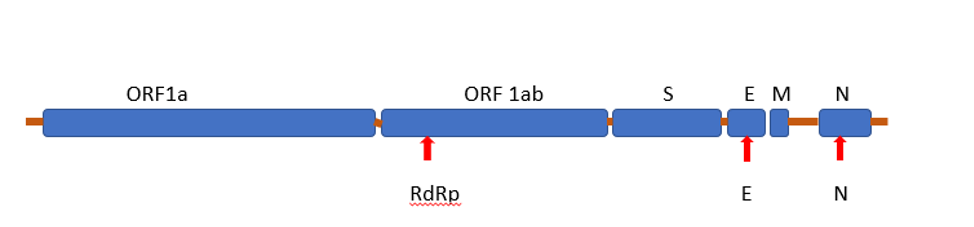
Figura 1 Amplicón diana del genoma del SARS-CoV-2. E: gen de la proteína de la envoltura; M: gen de la proteína de la membrana; N: gen de la proteína de la nucleocápside; ORF: marco de lectura abierto; RdRp: gen de la ARN polimerasa dependiente del ARN; S: gen de la proteína de la espiga.
La prueba QuantVirus Real-Time PCR Coronavirus (SARS-CoV-2) Detection Test es un ensayo de diagnóstico in vitro basado en qRT-PCR para la detección cualitativa de ARN viral de SARS-CoV-2.
Tres genes del SARS-CoV-2 incluyendo N, Orf1ab y E son el objetivo del ensayo qRT-PCR y los cebadores y sondas Taqman están diseñados en la región conservada de la región del genoma específica del virus SARS- CoV-2 para permitir la amplificación y detección sensible y específica del virus. El gen de la RNasa P humana se utiliza como control interno y de extracción para monitorizar la eficacia de la extracción del ARN viral y evaluar el ARN/ADN amplificable en las muestras a analizar.
Rendimiento del ensayo
El ensayo tiene un amplio rango dinámico como se muestra en la siguiente figura: Curva de amplificación de diluciones seriadas de 10 veces de plantillas que muestra el rango dinámico.
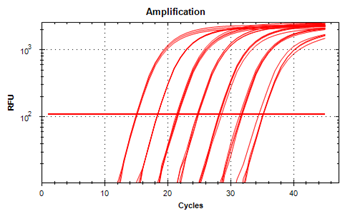
Resumen del rendimiento de la prueba QuantiVirus Real-Time PCR Coronavirus (SARS- CoV-2) Detection Test:
- Reproducibilidad intraensayo: Coeficiente de variación (%): < 3%
- Reproducibilidad del operador: Coeficiente de variación (%): < 2%
- Reproducibilidad entre instrumentos: Coeficiente de variación (%): < 6%
- Límite de detección del ensayo (LOD): 1 copia de ARN del virus o 100 copias por ml de muestra

