Oncogramme®: Hacia una medicina personalizada para todos los pacientes con cáncer

1 paciente - 1 tumor - 1 Oncogramme® - 1 tratamiento
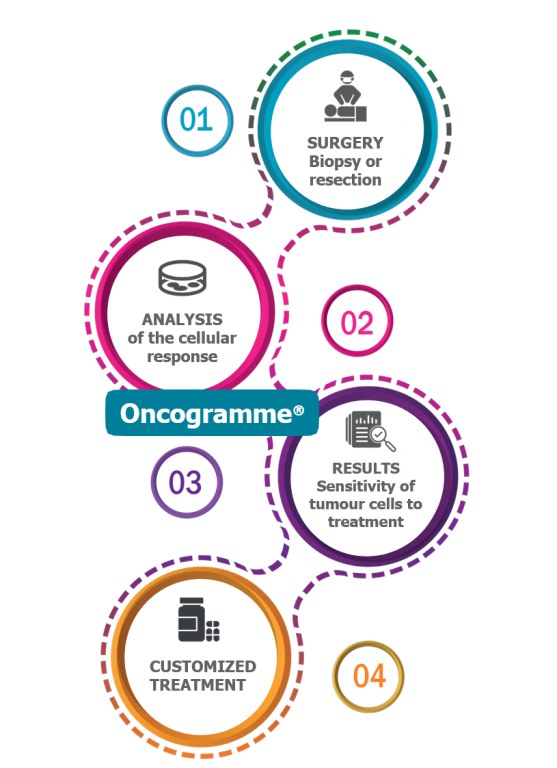
¿Qué es el Oncogramme®?
El Oncograma® predice la actividad anticancerígena de los medicamentos existentes probándolos directamente en el tumor operado de un paciente. Su eficacia se evalúa en una muestra de este tumor ex vivo, utilizando un enfoque comparable al del antibiograma.
Reconocimiento de la "Haute Autorité de Santé" francesa
En diciembre de 2019, Oncomedics recibió una decisión favorable de la Autoridad Nacional Francesa de Salud (HAS) a través del "Forfait Innovation" para el reembolso derogatorio del Oncogramme®.
Esta aprobación oficial de la Autoridad Nacional Francesa de la Salud recompensa tanto el alto nivel de innovación del Oncogramme® como su extraordinario impacto en el recorrido del paciente.
-
Marcado CE: Oncogramme® para mCRC cuenta con el marcado CE desde 2015
-
Oncomedics es una de las 13 empresas innovadoras que han recibido el "Forfait innovation" de HAS
-
En marzo de 2021, en el marco de la innovación Forfait, el Ministerio de Sanidad ha decidido reembolsar el Oncogramme®
Forfait Innovación
El "Forfait Innovation" es un reembolso temporal y derogatorio para facilitar el acceso temprano de los pacientes a tecnologías innovadoras (tecnologías sanitarias distintas de los medicamentos) en las primeras fases de desarrollo clínico.
La tecnología debe aportar pruebas de que es probable que su uso aporte beneficios significativos para la salud o reduzca los costes sanitarios.
¿Cómo personalizar el tratamiento?
Cada paciente con cáncer requiere una atención especial, que idealmente implica un tratamiento individualizado.
En la actualidad, la mayoría de los pacientes necesitan quimioterapia para combatir eficazmente el cáncer, pero ninguna prueba o biomarcador puede predecir la eficacia de los tratamientos o combinaciones de tratamientos. Las terapias dirigidas son una primera línea de defensa contra la enfermedad, pero no todas las personas pueden optar a ellas.
Basándose en un modelo similar al del antibiograma, nuestro laboratorio francés ha desarrollado y comercializado su propio test funcional. Las pruebas funcionales son una herramienta importante en la elección de la quimioterapia. Una revisión reciente demostró que las pruebas funcionales tenían una eficacia del 80%. (Bounaix Morand du Puch et al.,Theranostics. 2021 Sep 21;11(19): 9538-9556).
Desde 2016, los pacientes con cáncer colorrectal en estadio IV pueden beneficiarse del Oncogramme®: una solución única para personalizar la quimioterapia.
El Oncogramme® se optimizará próximamente para otros tipos de cáncer, como el de mama y el de ovario, con el fin de mejorar los tratamientos adaptándolos a cada paciente.
El cáncer colorrectal metastásico es la primera indicación del Oncogramme® DM-DIV con marcado CE.
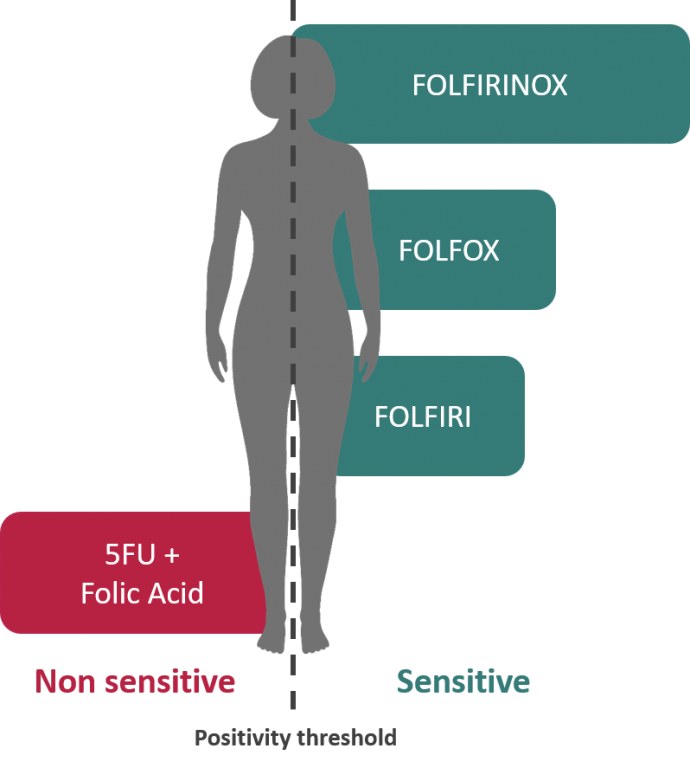
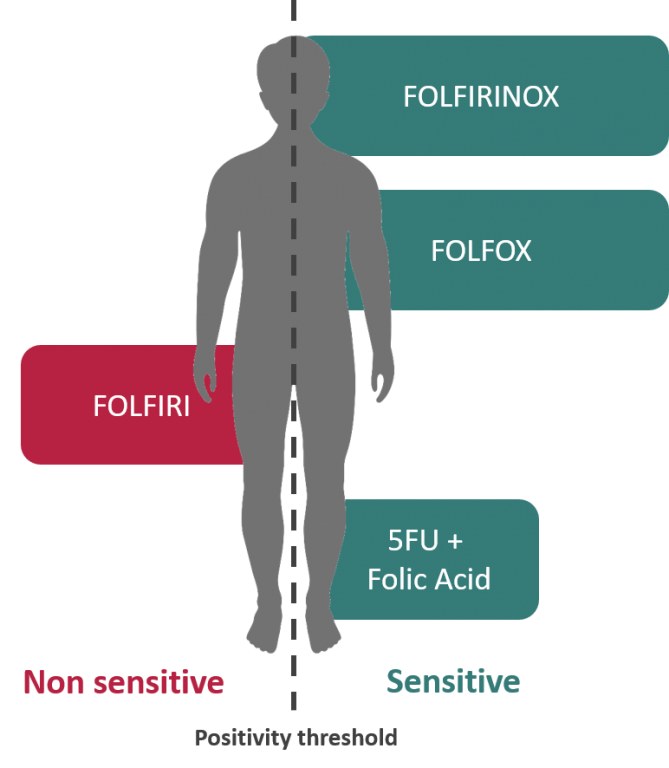
FOLFIRINOX : oxaliplatino, irinotecán, 5-fluorouracilo + ácido folínico
FOLFIRI : irinotecán, 5-fluorouracilo + ácido folínico
FOLFOX : oxaliplatino, 5-fluorouracilo + ácido folínico
FOLFIRI : irinotecán, 5-fluorouracilo + ácido folínico
FOLFOX : oxaliplatino, 5-fluorouracilo + ácido folínico
Los resultados de Oncomedics con el Oncogramme® en cáncer colorrectal metastásico sugieren una mayor probabilidad de respuesta al tratamiento de primera línea : 84 % (Bounaix Morand du Puch et al. J Transl Med. 2016 Jan 12;14: 10) en comparación con el 21% (5-FU solo) y el 48% (FOLFOX) en el mejor de los casos, según la bibliografía. (Pfeiffer et al. Onco Targets Ther. 2009; 2: 17-27) for the main consensus chemotherapeutic treatments.

Para más información:


